Prions
What they think they know
1. A prion is a proteinaceous infectious particle and can induce spongiform encephalopathies in a lot of animals, not only sheep (scrapie), cows (BSE) and humans (CJD and Kuru), but also rodents, minks (TME), elks (CWD) and probably pigs, chicken and some farmed fish.
2. BSE was first recognized in 1986, whereas Kuru had already been identified in 1957 (not the disease-causing agent itself, but a source where its residing in the harmful conformation. More about that below), scrapie is known for at least 200 years.
3. The harmless variant of the prion is called PrPc (Prion Particle cellular), the harmful one PrPsc (from scrapie).
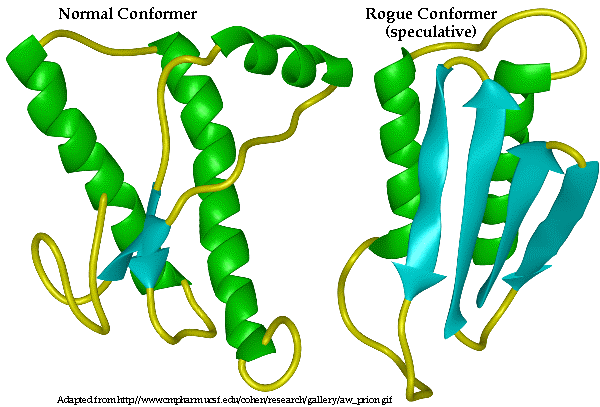
5. There are two ways to get the prion disease:
- Inheritable: about 10-15% of the CJD cases because one (or a few) out of the 750 codons is different as a result of a point mutation in the DNA: Leucine is substituted for Proline. Theyve found 18 of those mutations up till now. Another important codon appears to be nr 129, building in Methionine into the PrP-chain instead of Valine. About 37% is so-called homozygote for this codon 129, meaning that both helices need to have this defect ("MM"). At first they thought homozygosity was required for developing the disease, but recent results show that heterozygotes will die too, the only difference is the longer incubation period. 51% of the population is heterozygote (MV, one strand coding for the Methionine and one for Valine).
Reduced PrPc-gene expression delays the onset of the disease, whereas absence of the PrP gene makes the tested knockout mice fully resistant to infection. So production of PrPc is required for the PrPc to turn into PrPsc. On the other hand, excessive PrP production results in destruction of muscles and peripheral nerves. - Communicable: iatrogenic (e.g. via corneal transplantation, human growth hormone medicines), animal feed, food, and in some species like sheep also via blood.
What they suspect
I. Some prions cause disease quickly, while other prions (in the same species) have a longer incubation period. There is a tendency to believe that this may be dependent on the amino acid sequence: with one or a few other amino acids built in into the chain, one (or some) crucial parts of the prion may, or may not, be less stable hence more susceptible to changing the alpha-helix into the beta-sheet (i.e. PrPc into the harmful PrPsc). Tests were conducted with a PrP hybrid built of mice PrP-gene code and flanking human PrP code, built into and expressed in mice. A hybrid protein was produced. Then brain tissue of patients who died of CJD was introduced into the mice. Weird stuff was, that the transgenic mice became ill much more frequently and faster than did mice carrying a full human PrP gene (diverges from mice PrP at 28 position). This suggest that similarity in the supposed active region is important, as well as a "chaperone" molecule normally involved in folding nascent protein chains, recognized one or both mouse-derived regions of the PrPc.
II. PrPc is predominantly found on the surface of neurons, attached by a glycoinositol phospholipid anchor. What is its purpose there? (See also 4)
III. Ingested prions may be absorbed across the gut wall at Peyers patches. The resident bacteria may be facilitating a protective immune response (but others note that the white blood cells dont respond to prions at all). The lymphoid cells take up the prions by phagocytose, so that the prions can travel to lymphoid nodes, spleen and tonsils and probably replicate there. "Eventually they gain access to a nerve" (How?) and can propagate or travel via the axon and spinal cord up to the brain. Either glial cells or neural cells can propagate the disease independently. And the fact that intercerebral injection of PrPsc alone doesnt cause pathology means that "cells must be making PrP for a pathological result". I dont think thats a good conclusion, instead of "must" it should be at least "may" (and see point II of this section): the fact that theres no known physiological function doesnt mean that it doesnt exist. If you dont know where, what and how exactly to test (as is the situation trying to solve the mystery around prions; "standard" infectious particles are normally micro-organisms or viruses, at least some DNA or RNA involved, never a parasiting protein) it is not likely youre going to find it easily.
IV. Results tend to be in the direction that the conformation change apparently happens on a membrane in the cell interior. Apparently in neurons. They accumulate in the lysosomes in the cell, which will burst and then infect other cells. The plaques that are sometimes mentioned are not always observed, nor that it disturbs intracellular processes per se. See also next point V.
V. There is a suspicion of the existence of prion "strains", multiple possible conformations of prions. This is based on the idea: hey, weve found two conformations, if there are two, it makes sense if there are more, and that would explain the different results seen (disease symptoms, location of the prions in the body.)
Hamster prion conformations
What they really dont know
(or I just couldnt find the answer)
A. All research seems to be directed towards the harm the prions can do, but the PrPc variant is produced in a lot of animals, which indicates that the gene sequence is evolutionary old. Does this mean that PrPc is also beneficial to the body? Why is it produced in the first place? Involved in synaptic functions? (See points II and 4)
B. After transcription and translation of the PrPc, there are extensive post-translational modifications (mainly conformational changes) made to the protein, meaning that the protein is realtively advanced and evolved. How does that happen? What exactly is changed, and what is the sequence of these events?
C. Pigs, chicken, sheep and farmed fish all have been exposed to BSE-contaminated feed. But what is the difference in the DNA and tertiary structure of the prions? And I dont mean the answer that there are 30 different positions between bovine and human prions, or the 7 between sheep and cows, I know that. But some parts of the protein are less important for functioning than others. Are those differences in supposed active sites of the prion? The disease pattern in humans tend to be more similar to sheep, so do sheep infect humans more easily? (Side note: currently there doesnt seem to be a so-called consistent correlation between countries herding and eating a lot of sheep and CJD, but see also next point) And if the transmission pattern thought to be similar, what about blood transfusions? (This is being investigated now)
D. If the sheep fed with BSE-contaminated meat can pass on their prions to mice, can they do that to humans too? And if yes, are the symptoms similar to scrapie, or BSE, or something else we dont know yet? I dont want to be highly suggestive, but take a disease like Alzheimer: physiologically the brain gets porous in a relatively similar way like CJD, leading to the question: Are they both related to prions or prion-like molecules?
E. PrPsc catalyses the conformation (only? And alone?) change from the alpah-helix in PrPc into the beta-sheet in PrPsc. But initially only "harmless" PrP is produced. There must have been a first flipping prion. Produced by the body itself, e.g. that the post-translational machinery doesnt work properly anymore? Was the protein PrPc getting old and changed conformation? To which extend is it possible for a PrPsc from say a cow to induce the flipping of PrPc in humans (or whatever other test animal) (see point I)?
F. They dont know the process during the conformation change.
Still a lot of uncertainties, like with most natural science research. But thats also its charm - well, for people who like to dig into the topic of course. If anyone knows more, or even has answers on the above questions, I'm interested.
Most information is from articles from New Scientist, Scientific American and Nature. A well-written resource that also provides insight in "their search for truth" is the article at http://www.nmia.com/~mdibble/prion.html. Lots of gory details also can be found on the mad cow website.